Enzymes: Classification, Mechanism of Action and Applications
Enzymes are the biocatalysts. A catalyst is defined as a substance that increases the velocity or rate of a chemical reaction without itself undergoing any change in the overall process.
Enzymes may be defined as biocatalysts synthesized by living cells. They are protein in nature (exception – RNA acting as ribozyme), colloidal in nature and thermolabile in character, and highly specific in their action.Enzymes are sometimes considered under two broad categories:
- (a) Intracellular enzymes – They are functional within cells where they are synthesized. Examples are, Hexokinase, DNA Ploymerase, ATP synthatase etc.
- (b) Extracellular enzymes – These enzymes are active outside the cell; all the digestive enzymes belong to this group. Examples are: Amylase, Pepsin, Cellulase, Lipase etc
Classification of Enzymes
As per the IUB system of enzyme classification has been in force. Enzymes are divided into six major classes (in that order). Each class on its own represents the general type of reaction brought about by the enzymes of that class. One can remember these categories by the word O T H L I L.
- 1. Oxidoreductases: Enzymes involved in oxidation-reduction reactions.
- 2. Transferases: Enzymes that catalyse the transfer of functional groups.
- 3. Hydrolases: Enzymes that bring about hydrolysis of various compounds.
- 4. Lyases: Enzymes specialised in the addition or removal of water, ammonia, CO2 etc.
- 5. Isomerases: Enzymes involved in all the isomerization reactions.
- 6. Ligases: Enzymes catalysing the synthetic reactions where two molecules are joined and ATP is used.
Chemical Nature of Enzymes
Enzymes are biological macromolecules made primarily of proteins. They are composed of long chains of amino acids folded into complex 3D shapes and after many folding that aamino acid chain produce quaternary structure.
This specific shape is essential for their catalytic activity as due to this folding active and binding site are generated. These sites are essential for enzyme activities and for binding of cofactors and coenzymes.The complete enzyme has a protein and non-protein part. The non-protein part is essential for enzymatic activity.
- Apoenzyme: The protein part of an enzyme (inactive alone).
- Cofactor: The non-protein part (can be a metal ion like Mg²⁺, Fe²⁺, Zn²⁺).
- Coenzyme: The non-protein part (an organic molecule e.g., NAD⁺, FAD, coenzyme A)..
- Holoenzyme: The complete, active enzyme (apoenzyme + cofactor).
- Prosthetic Group: The cofactor or coenzyme, which is strongly attached with apoenzyme.

Enzyme Sites
Enzyme has some sites on it, which areActive Site: The active site is the small but specific region of an enzyme where the substrate binds by weak noncovalent bonds and the chemical reaction takes place. In other words, it is the site which is responsible for conversion of substrate into product. Generally, the active site possesses a substrate binding site and a catalytic site.
Catalytic Site: is a part of active site, Coenzymes and Cofactors are bind at this site, coenzymes and cofactors are essential for activity of some enzyes.
- Substrate Binding Site: this site is also a part of catalytic site, where the substrate can bind and by the action of catalytic site, converted into product.
Allosteric Site: An allosteric site is a specific region on an enzyme, separate from the active site, where a molecule binds and changes the enzyme’s activity by altering its shape or conformation. At this site any chemical or any inhibitor can bind, which affect or reguate the enzyme activity
Mechanism of Enzyme Activity
Enzymes are biological catalysts that speed up the biochemical reactions by lowering the activation energy required for the reaction to occur.
Activation energy is the minimum amount of energy needed to convert reactants into products. Without enzymes, many biochemical reactions would proceed too slowly due to high activation energy. Enzymes function by binding to the substrate (the reactant molecule) and forming an enzyme-substrate complex. This interaction helps to properly orient the substrate molecules, weaken specific chemical bonds, and stabilize the transition state (Enzyme substrate complex), which is the high-energy intermediate stage of the reaction.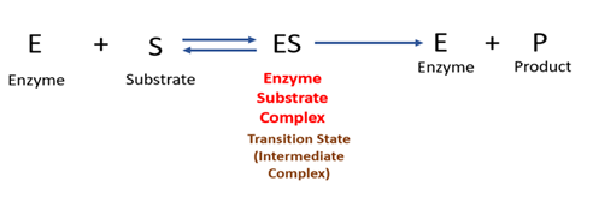
 Enzymes lowers the activation energy of a reaction by providing an easier pathway for the reaction to occur.
Activation energy is the minimum energy required for reactant molecules to reach the transition state (Enzyme - substrate complex, in this state, old chemical bonds are partially breaking and new bonds are partially forming, due to this the substrate is converted into product.) and get converted into products.
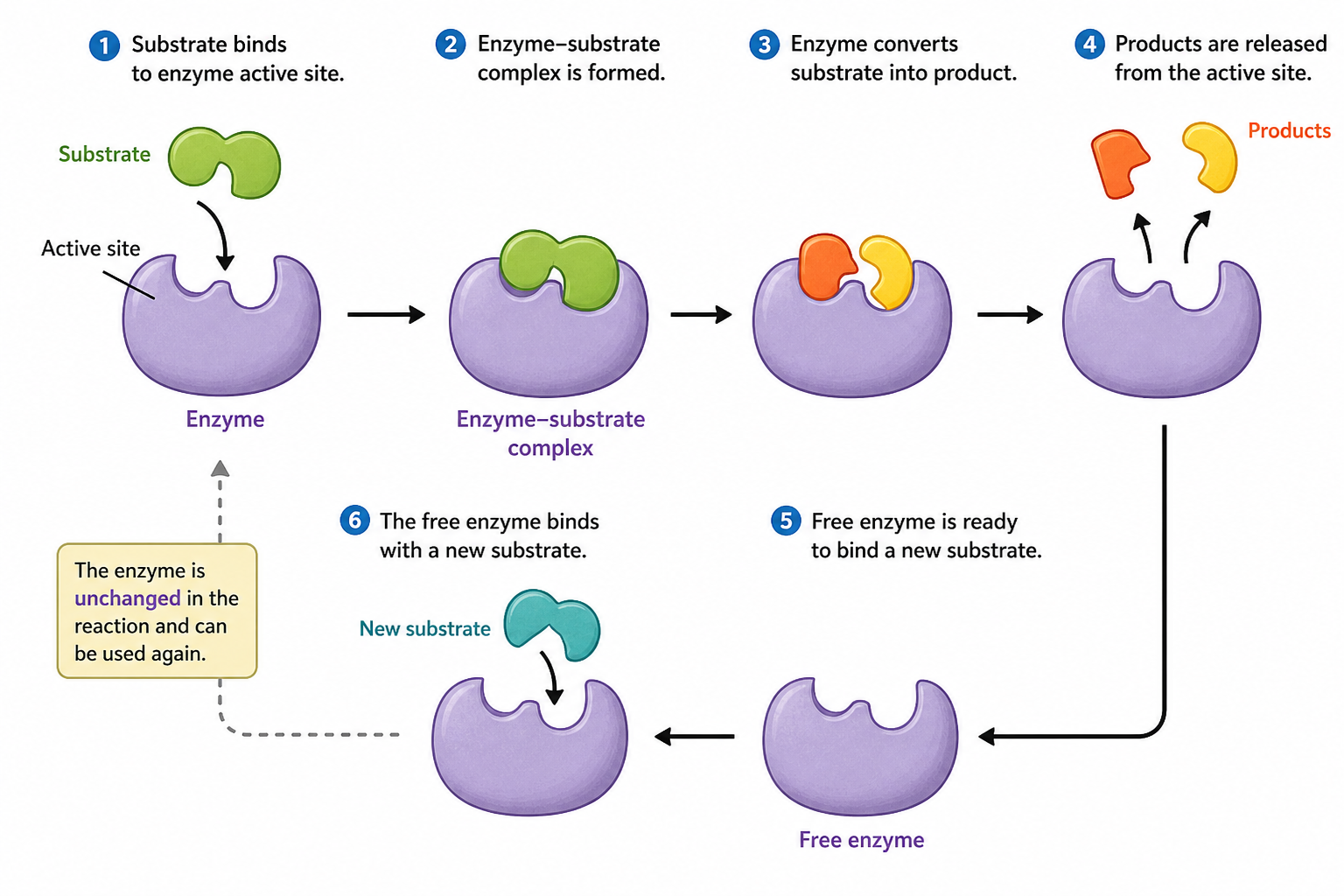
Normally, reactant molecules may not have enough energy or the correct orientation to react efficiently, so the reaction occurs very slowly. When a substrate binds to the active site of an enzyme, an enzyme–substrate complex is formed. The enzyme then helps the reaction to perform easily through several ways:
Enzymes lowers the activation energy of a reaction by providing an easier pathway for the reaction to occur.
Activation energy is the minimum energy required for reactant molecules to reach the transition state (Enzyme - substrate complex, in this state, old chemical bonds are partially breaking and new bonds are partially forming, due to this the substrate is converted into product.) and get converted into products.
Normally, reactant molecules may not have enough energy or the correct orientation to react efficiently, so the reaction occurs very slowly. When a substrate binds to the active site of an enzyme, an enzyme–substrate complex is formed. The enzyme then helps the reaction to perform easily through several ways:
- It brings the reacting molecules close together.
- It properly orients the molecules for reaction.
- It weakens certain chemical bonds in the substrate.
- It stabilizes the transition state of the reaction.

Enzyme Inhibition
Enzyme inhibition refers to the decrease in enzyme activity due to the interaction with specific molecules known as inhibitors. This inhibition can be classified as either reversible or irreversible based on the nature of the inhibitor’s interaction with the enzyme.
- Reversible Inhibition: In which the inhibitor binds to the enzyme through non-covalent interactions such as hydrogen bonds, ionic bonds, or hydrophobic forces. This binding is temporary and can be reversed, allowing the enzyme to regain its activity once the inhibitor is removed. Reversible inhibitors are further categorized into competitive, non-competitive, uncompetitive, and allosteric types.
- Competitive: Competitive inhibition occurs when an inhibitor competes with the substrate for the enzyme’s active site.The inhibitor binds to the active site and blocks the substrate from attaching. As a result, the enzyme cannot form the product. There is a competition between substrate and inhibitor to bind to the active site because the inhibitor has a similar structure to the substrate, so both can fit into the same site, that is why they both competes to each other to bind with active site.This type of inhibition raises the apparent Km (Michaelis constant) but does not affect the maximum reaction velocity (Vmax).
- Non-Competitive: In non-competitive inhibition, the inhibitor binds to a binding site, which is different from the active site—on either the free enzyme or the enzyme-substrate complex. This binding changes the enzyme’s conformation, reducing its catalytic efficiency regardless of substrate concentration. As a result, non-competitive inhibition decreases Vmax, but does not alter Km, because the substrate can still bind with the same affinity but not converted to product.
- Uncompetitive: In this, the inhibitor binds only to the enzyme-substrate complex, not to the free enzyme. This binding typically occurs at a site other than the active site. In uncompetitive inhibition, the inhibitor binds to the enzyme–substrate (ES) complex and forms an inactive ESI (Enzyme-Substrate-Inhibitor Complex) complex. This binding stabilizes the enzyme–substrate complex, so the enzyme cannot convert the substrate into product efficiently. Uncompetitive inhibition reduces both Vmax and Km, and unlike competitive inhibition, it cannot be reversed by simply increasing substrate concentration.
- Irreversible Inhibition: On the other hand, irreversible inhibition involves a permanent inactivation of the enzyme. Irreversible inhibitors typically form covalent bonds with the enzyme, often targeting the active site or a critical functional group necessary for enzymatic activity. This modification permanently alters the enzyme’s structure, rendering it non-functional. Since the enzyme cannot be reactivated, the cell must synthesize new enzyme molecules to restore function.


Diagnostic Applications of Enzymes
The diagnostic applications of enzymes refer to the use of specific enzymes as biomarkers to detect, monitor, or confirm diseases. Because enzymes are often released or altered in concentration due to tissue damage or disease, they serve as important indicators of underlying conditions.

Therapeutic Applications of Enzymes
Enzymes are used in treatment of various diseases:
